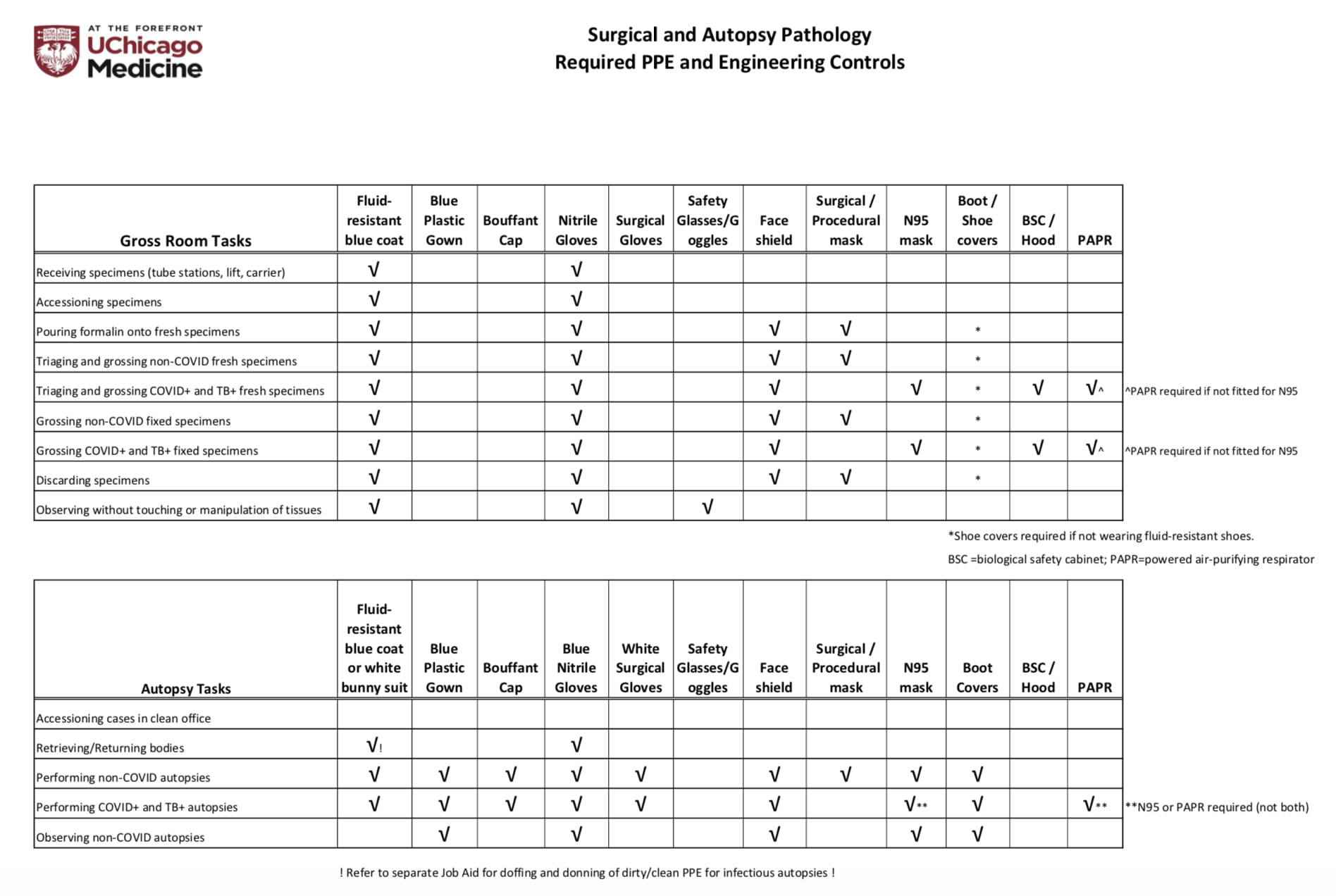
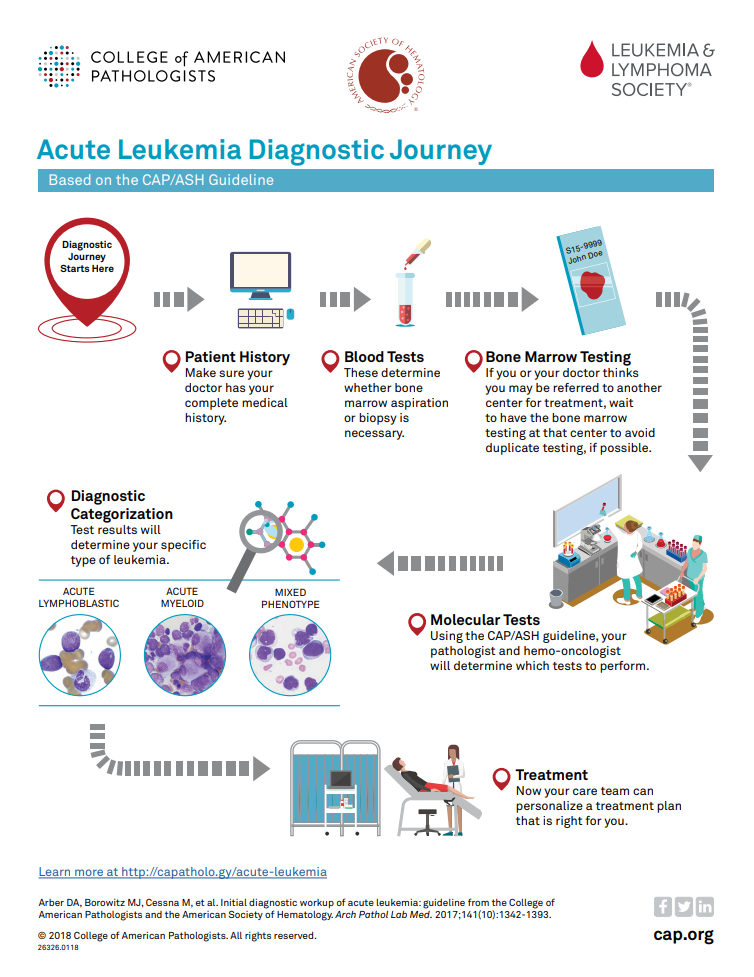
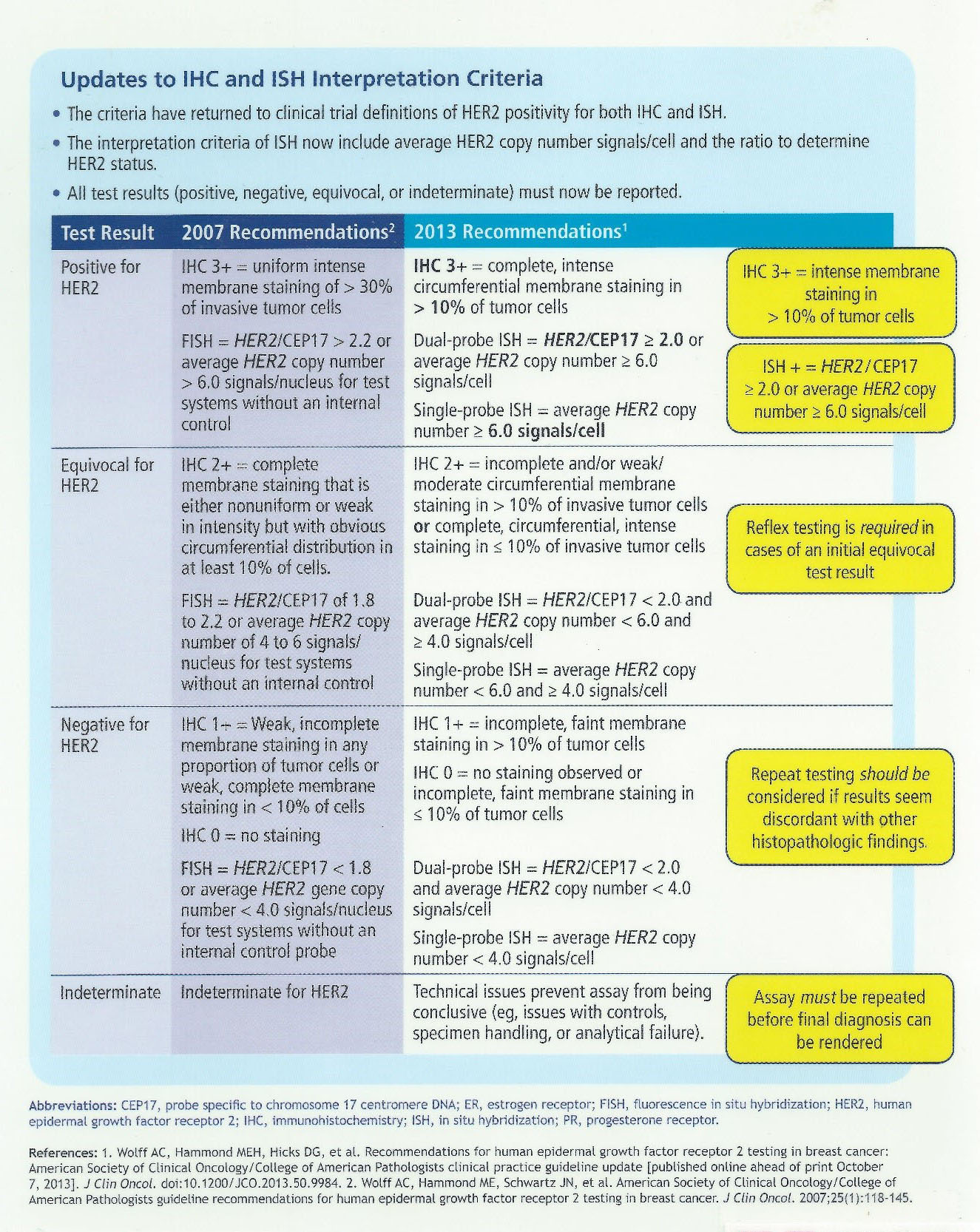
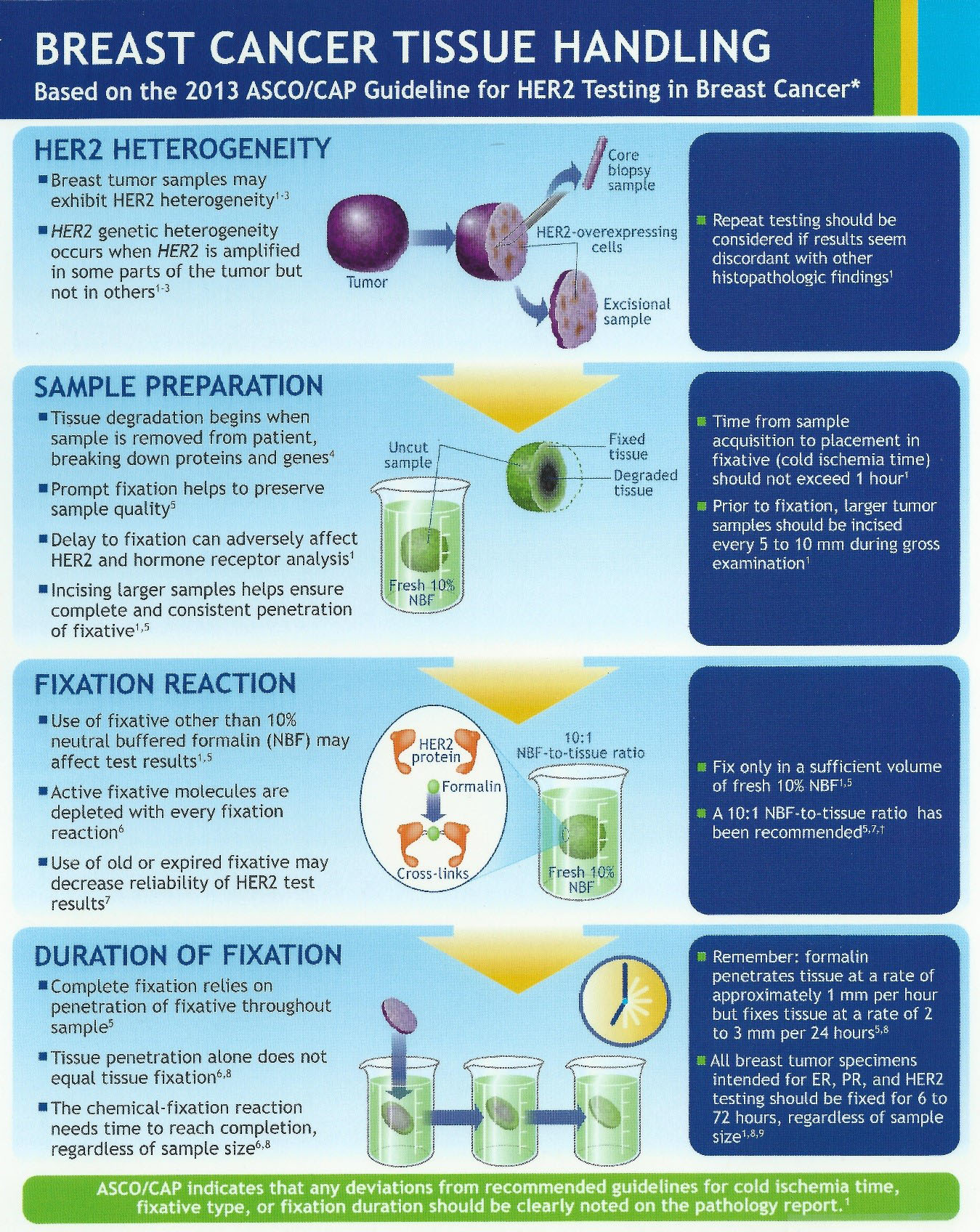
DATE: TO: Reference Lab Clients SUBJECT: CAP-NSH Uniform Labeling Requirements for Blocks and Slides in Surgical Pathology The C

AAPA Student Committee - 5-15 % of the ASCP BOC exam covers laboratory operations. Don't miss out on these questions; here is a quick review on CAP retention guidelines as you prepare
Protocol for the Examination of Resection Specimens From Patients With Invasive Carcinoma of the Breast

Medicina | Free Full-Text | Awareness of Placental Pathologic Examination Criteria and Utilization of Pathology Reports among Obstetricians

PDF) American Society of Clinical Oncology/College of American Pathologists Guideline Recommendations for Immunohistochemical Testing of Estrogen and Progesterone Receptors in Breast Cancer

Reporting Guidelines for Clinical Laboratory Reports in Surgical Pathology ( Cap Laboratory Improvement Programs) (Clinical Report) on Apple Books
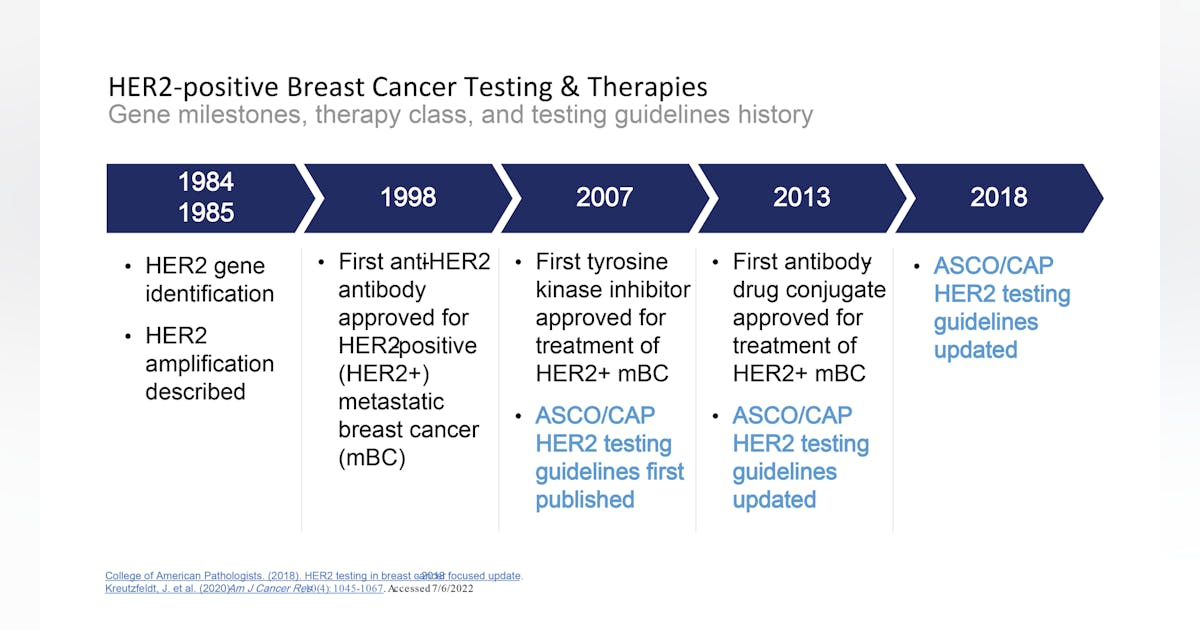
Breast cancer biomarkers, and a new clinical category for HER2 expression | Medical Laboratory Observer