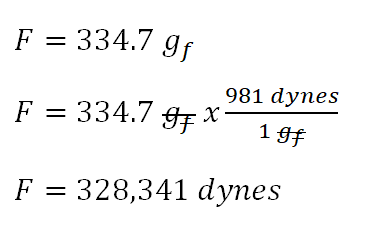Activity 1: solve for size and speed of a drop.. Useful units for calculations Remember: dyne = 1 g cm/s 2 (force in cm-g-s units) = Newtons. Using. - ppt download

Convert the folowing (a) 5 dyne/cm^2 into N/m^2 (b) 10 g/cm^3 into Kg/m^3 - Physics - Units And Measurements - 12979011 | Meritnation.com

How to convert 1 N (newton) into 10^5 dyne by Dimensional analyses fully explained.......... - YouTube

The Undertale fandom wiki says Undyne's name came from "Undine, a womanly water nymph from Greek lore." But what if its also a play on this? A "dyne" is a very very

One - gram mole of nitrogen occupies 2 × 10^4 cc at a pressure of 10^6 dynes/cm^2 . The average energy of a nitrogen molecule (in erg) will be: (Avogadro's number = 6 × 10^25 )

SOLVED:If at a pressure of 10^6 dyne / cm^2, one gram mole of nitrogen occupies 2 ×10^4 c.c. volume, then the average energy of a nitrogen molecules in erg is : (Given: